MR SPECTROSCOPY IN EPILEPSY
 Epilepsy, the condition of recurrent
seizures, is a relatively common neurological
disorde. A multitude of etiologies cause epilepsy,
including tumors, developmental abnormalities,
febrile illness, trauma, or infection. However, not
infrequently, the cause is unknown. Many patients
with epilepsy can be successfully treated
pharmacologically, but when medical management fails
to adequately control seizure activity, surgical
resection of the epileptogenic tissue may be
considered. For surgery to be successful, seizures
must be of focal onset from a well-defined location.
It has been estimated that up to 10% of patients
with epilepsy are medically intractable, of whom
approximately 20% may be candidates for surgical
treatment.
Epilepsy, the condition of recurrent
seizures, is a relatively common neurological
disorde. A multitude of etiologies cause epilepsy,
including tumors, developmental abnormalities,
febrile illness, trauma, or infection. However, not
infrequently, the cause is unknown. Many patients
with epilepsy can be successfully treated
pharmacologically, but when medical management fails
to adequately control seizure activity, surgical
resection of the epileptogenic tissue may be
considered. For surgery to be successful, seizures
must be of focal onset from a well-defined location.
It has been estimated that up to 10% of patients
with epilepsy are medically intractable, of whom
approximately 20% may be candidates for surgical
treatment.
 Traditionally, scalp electroencephalography (EEG)
and often invasive (subdural grid or depth
electrode) EEG are used to identify the
epileptogenic regions of the brain, but increasingly
magnetic resonance imaging (MRI), positron emission
tomography (PET), ictal single photon emission
computed tomography (SPECT), and, more recently,
magnetoencephalography (MEG) are also used.
Traditionally, scalp electroencephalography (EEG)
and often invasive (subdural grid or depth
electrode) EEG are used to identify the
epileptogenic regions of the brain, but increasingly
magnetic resonance imaging (MRI), positron emission
tomography (PET), ictal single photon emission
computed tomography (SPECT), and, more recently,
magnetoencephalography (MEG) are also used.
 MRI is the modality of choice for identifying brain
tumors, cortical malformations, infectious and other
causes of epilepsy. In mesial temporal sclerosis
(MTS), the most common abnormality in patients with
temporal lobe epilepsy, MRI typically shows
hippocampus volume loss, with abnormal signal
intensity on T2-weighted images, which corresponds
histologically to neuronal loss and gliosis.
Sensitivity of MTS detection may be increased by
performing careful, quantitative T2 measurements
from multiple echo data acquisitions, or by using
the CSF-suppressed FLAIR sequence. Quantitative
volume measurements more reliably detect small
changes in hippocampal volume and are generally
preferable, particularly when atrophy may be subtle.
Lateralization of seizure focus in patients with
temporal lobe epilepsy has been reported to be over
90% efficient with volumetric analysis of
hippocampal and amygdaloid formations using
high-resolution 3D scans. While these studies show
that MRI is a sensitive tool for the detection of
MTS, the clinical significance of these findings
should be carefully considered. First, many
published studies have been performed in
retrospectively selected patients who were already
candidates for epilepsy surgery by other criteria,
such as EEG. This may increase sensitivity and
specificity by excluding patients who might have
negative MRI findings, or who are “complicated”
cases. Second, a significant number of patients will
have symmetric hippocampi (either no atrophy or
bilateral atrophy), and yet still have successful
seizure control after surgery, indicating that
bilateral sclerosis is not necessarily a
contra-indication for surgery. Third, longer-term
follow-up post-surgery is often not reported; one
study found seizure-free outcome in 70–80% of
patients 1 year after surgery, but by 5 years this
number had fallen to 50–60%. Interestingly, relapse
only occurred in the patients who originally
presented with hippocampal atrophy. Collectively,
these studies demonstrate that MRI is a valuable
tool for the evaluation of patients with epilepsy,
but that it also has limitations. For this reason,
other imaging studies are often considered for the
evaluation of epilepsy patients, particularly
“functional” techniques that measure blood flow and
metabolism, such as SPECT and PET.
MRI is the modality of choice for identifying brain
tumors, cortical malformations, infectious and other
causes of epilepsy. In mesial temporal sclerosis
(MTS), the most common abnormality in patients with
temporal lobe epilepsy, MRI typically shows
hippocampus volume loss, with abnormal signal
intensity on T2-weighted images, which corresponds
histologically to neuronal loss and gliosis.
Sensitivity of MTS detection may be increased by
performing careful, quantitative T2 measurements
from multiple echo data acquisitions, or by using
the CSF-suppressed FLAIR sequence. Quantitative
volume measurements more reliably detect small
changes in hippocampal volume and are generally
preferable, particularly when atrophy may be subtle.
Lateralization of seizure focus in patients with
temporal lobe epilepsy has been reported to be over
90% efficient with volumetric analysis of
hippocampal and amygdaloid formations using
high-resolution 3D scans. While these studies show
that MRI is a sensitive tool for the detection of
MTS, the clinical significance of these findings
should be carefully considered. First, many
published studies have been performed in
retrospectively selected patients who were already
candidates for epilepsy surgery by other criteria,
such as EEG. This may increase sensitivity and
specificity by excluding patients who might have
negative MRI findings, or who are “complicated”
cases. Second, a significant number of patients will
have symmetric hippocampi (either no atrophy or
bilateral atrophy), and yet still have successful
seizure control after surgery, indicating that
bilateral sclerosis is not necessarily a
contra-indication for surgery. Third, longer-term
follow-up post-surgery is often not reported; one
study found seizure-free outcome in 70–80% of
patients 1 year after surgery, but by 5 years this
number had fallen to 50–60%. Interestingly, relapse
only occurred in the patients who originally
presented with hippocampal atrophy. Collectively,
these studies demonstrate that MRI is a valuable
tool for the evaluation of patients with epilepsy,
but that it also has limitations. For this reason,
other imaging studies are often considered for the
evaluation of epilepsy patients, particularly
“functional” techniques that measure blood flow and
metabolism, such as SPECT and PET.
 Epilepsy has been extensively studied by PET since
the early 1980s, the majority of the studies using
18F-fluorodeoxyglucose (FDG) to measure glucose
metabolism. Interictally, glucose uptake is reduced
compared to normal brain, while ictally increases in
uptake may be observed. The hypometabolic region is
usually larger on interictal PET scans than the
electrically defined volume of pathology. Seizure
foci also have been found to be associated with
changes in cerebral perfusion, which can be
monitored with oxygen-15 PET, SPECT, or perfusion
MRI. In one study, PET and MRI were determined to be
comparable in terms of ability to detect
abnormalities in patients with temporal lobe
epilepsy, but PET had better concordance than MRI
with the EEG localization.
Epilepsy has been extensively studied by PET since
the early 1980s, the majority of the studies using
18F-fluorodeoxyglucose (FDG) to measure glucose
metabolism. Interictally, glucose uptake is reduced
compared to normal brain, while ictally increases in
uptake may be observed. The hypometabolic region is
usually larger on interictal PET scans than the
electrically defined volume of pathology. Seizure
foci also have been found to be associated with
changes in cerebral perfusion, which can be
monitored with oxygen-15 PET, SPECT, or perfusion
MRI. In one study, PET and MRI were determined to be
comparable in terms of ability to detect
abnormalities in patients with temporal lobe
epilepsy, but PET had better concordance than MRI
with the EEG localization.
Worse surgical outcome was also associated with
hypometabolism which extended beyond the region of
the temporal lobe. MRI and PET imaging are usually
done interictally for logistical reasons.
 The main advantage of SPECT, despite having lower
resolution and being less quantitative than PET, is
that SPECT can be performed ictally, i.e. the SPECT
tracer is injected while (or just after) the patient
experiences a seizure. Ictal SPECT may provide
unique information about the location of the
epileptic focus.
The main advantage of SPECT, despite having lower
resolution and being less quantitative than PET, is
that SPECT can be performed ictally, i.e. the SPECT
tracer is injected while (or just after) the patient
experiences a seizure. Ictal SPECT may provide
unique information about the location of the
epileptic focus.
 All together, these studies demonstrate the
additional value of flow and metabolic based imaging
studies, in addition to structural anatomic scans,
for the evaluation of patients with non-lesional
epilepsy.
All together, these studies demonstrate the
additional value of flow and metabolic based imaging
studies, in addition to structural anatomic scans,
for the evaluation of patients with non-lesional
epilepsy.
 MR spectroscopy in epilepsy
MR spectroscopy in epilepsy
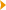 31P MRS
31P MRS
The use of MR spectroscopy for the metabolic
evaluation of animal models of epilepsy was first
investigated by the Yale group in the early 1980s.
At that time, 31P was the most widely used nucleus
for in vivo MR studies of the brain, although some
proton MRS studies were also performed. Generally,
status epilepticus is associated with reductions of
high-energy phosphates (nucleotide triphosphates
(NTP) and phosphocreatine (PCr)), increases in
low-energy phosphates (inorganic phosphate (Pi)),
and cerebral acidosis as determined by the chemical
shift of the Pi peak. However, not all studies
report these findings; in an interictal cortical
spike focus in the rat, no significant 31P MRS or
MRI changes were reported. After these initial
results in animal models, the use of 31P MRS in
humans with seizure disorders was reported in the
late 1980s and early 1990s. Interictally, seizure
foci were found by 31P MRS to be alkaline, and this
was proposed as a means of lateralization of the
seizure foci, although this finding was not
reproduced subsequently. 31P MR spectroscopy of
infants experiencing status epilepticus showed a
decrease in the PCr/Pi ratio, and in general, in
most forms of adult epilepsy, the most common
finding (ictally or interictally) appears to be
bioenergetic impairment (i.e. reduced ratios of
PCr/Pi and/or PCr/ATP). In one study using 31P MRS
at high field (4.1 T) in a group of 30 patients, 31P
MRS successfully lateralized temporal lobe epilepsy
in 70–73% using either PCr/Pi or ATP/Pi ratios, a
rate that was actually higher than that achieved
with MRI in the same study. An example of a 31P MRS
of a 2-year-old child with Lennox–Gastaut syndrome,
before and after initiation of the “ketogenic diet”
for seizure control, a small but noticeable increase
in PCr can be determined.
 However, the relatively coarse spatial resolution
and low sensitivity of MRS (≈30 cm3 voxel
size for human brain studies at 1.5 T) limit the
application of this technique to rather large focal
abnormalities or diffuse brain pathologies. The
technique does not appear to be able to map the
extent of epileptogenic tissue because of its low
spatial resolution. Finally, it is not particularly
widely available since appreciable hardware
modifications are required on most MRI scanners as a
result of the lower resonant frequency
However, the relatively coarse spatial resolution
and low sensitivity of MRS (≈30 cm3 voxel
size for human brain studies at 1.5 T) limit the
application of this technique to rather large focal
abnormalities or diffuse brain pathologies. The
technique does not appear to be able to map the
extent of epileptogenic tissue because of its low
spatial resolution. Finally, it is not particularly
widely available since appreciable hardware
modifications are required on most MRI scanners as a
result of the lower resonant frequency
of the 31P nucleus. For all of these reasons, there
have been many more proton MRS studies of epilepsy
than 31P, although with high-field (i.e. 3 T and
above) MRI systems becoming increasingly available,
there is still some interest in using 31P to
investigate the biochemical processes occurring in
patients with epilepsy.
 High-field 31P MRS offers higher sensitivity which
results in better spatial resolution (~ 6–12 cm3
in 40–50 min scan time) compared to lower fields.
High-field 31P MRS offers higher sensitivity which
results in better spatial resolution (~ 6–12 cm3
in 40–50 min scan time) compared to lower fields.
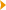 Proton MRS
Proton MRS
Proton spectroscopy has a considerable sensitivity
advantage compared to 31P which allows significantly
better spatial resolution, and can be used on most
MRI scanners without hardware modifications. Over
the last several years, therefore, most spectroscopy
studies of human epilepsy have utilized the proton
nucleus. The first published study involved two
patients with
Rasmussen’s syndrome (both of whom
had abnormal MRI scans). Both patients showed
decreased N-acetyl aspartate (NAA), and the single
patient who had seizures during spectral acquisition
showed increased lactate. Since the NAA signal
is believed to originate from neuronal cells,
the
reduction in NAA has been attributed to neuronal
loss within the seizure focus, which is also a
common
histological finding. Increased lactate in
patients
who are experiencing active seizures is consistent
with
the hypermetabolism observed in ictal FDG-PET scans,
indicating that the increased glucose uptake is at
least
partly metabolized anaerobically to lactate, as
opposed
to the normal path through pyruvate to the TCA
cycle.
Ictal, or early post-ictal (up to about 6 h)
elevations
of lactate have therefore been found to be useful in
the
identification of seizure foci. However, most
spectroscopy
studies of epilepsy are performed interictally
(where lactate is not normally observed), and the
most
universal finding associated with seizure foci is a
decrease
in NAA, either measured quantitatively or as a
ratio
to creatine, ratio to choline, or both. In
some cases, increases in choline may also be
observed,
perhaps as the result of gliosis or neoplastic
proliferation,
since glial cells are believed to have high choline
levels.
In short echo time spectroscopy, lower levels of
glutamate and glutamine (Glx) than in control
subjects
have also been reported in patients with hippocampal
sclerosis. This is consistent with lower
glutamate
(the major component of the “Glx” peak) in
association
with neuronal loss – in an in vitro study of
temporal lobe specimens from patients undergoing
epilepsy surgery, it was found that Glu gave an
excellent
correlation with NAA, with both Glu and NAA
showing trends for negative correlations with
hippocampal
neuronal counts. Consistent with this, an
MRSI study in temporal lobe epilepsy also found
lower
levels of Glu in patients with temporal lobe
epilepsy:
lower in the ipsilateral temporal lobe, but lower
than
healthy controls in the contralateral temporal lobe
as
well. Finally, in vitro NMR spectroscopy studies
of perchloric acid extracts of gliotic hippocampal
tissue
have also shown increased myo-inositol and decreased
glutamate, consistent with gliosis and neuronal
loss.
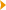 Temporal lobe epilepsy
Temporal lobe epilepsy
In early published studies of patients with temporal
lobe epilepsy (TLE), reductions of NAA in the
affected
hippocampus were found in 100%- 88%
(note 40% of the cases had bilateral reductions, so
that
lateralization was obtained in 60% of cases,
90%, 100%, and 100% of cases studied. An
example of proton MRS in a patient with unilateral
mesial temporal sclerosis, showing decreased NAA, is
depicted in Figure 1. Generally, sample sizes in
these
studies were in the range between 10 and 25
subjects,
and they most likely contained carefully
pre-selected
cases with clear-cut abnormalities on other
modalities.
Larger and more recent studies have reported more
variable success rates in terms of lateralizing
abnormalities
in patients with temporal lobe epilepsy; in one
MRSI study in 50 patients, success of localization
(based on neuroradiological interpretation of
spectra)
varied from 62% to 76%, while in another the
NAA/Cr ratio only successfully lateralized 18 of 40
cases (45%); however, cases which were lateralized
by
MRS had excellent surgical outcomes. In a study
of
100 cases (using MRSI and volumetric MRI), MRSI was
found to correctly localize 86% of cases, very
similar to volumetric MRI and EEG localization
rates.
Despite published studies such as these with high
success rates, MRS and/or MRSI have had relatively
little clinical impact over the last few years for
presurgical
evaluation of epilepsy patients. There are probably
several reasons for this.
(1) Research studies typically pre-select
well characterized
patients to study, who are
somewhat different from the general epileptic
population, and not typical of the “difficult” cases
that may be referred for special MRS studies.
(2) The spectroscopic changes are subtle, so while
group
studies may show statistically significant
differences,
decision-making confidence in individual patients
may be low.
(3) The site of abnormality in many patients, the
anterior mesial temporal lobe, is often in a region
of poor field homogeneity because of magnetic
susceptibility effects from adjacent paranasal
sinuses
and mastoids, leading to poor quality spectra that
are difficult to interpret.
(4) Metabolic abnormalities may be bilateral, even
in
patients with unilateral MTS or who have good
surgical outcome following unilateral temporal
lobectomy (i.e. seizure free after 1 year – class 1
on
the Engel surgical outcome scale).
(5) In many patients, MRI and MRS may be
concordant, which, while improving confidence in
the diagnosis, may not warrant the performance of
the MRS study in addition to conventional MRI.
Points (4) and (5) above warrant some extra
discussion
below.
 Diffuse metabolic abnormalities
in temporal lobe epilepsy
Diffuse metabolic abnormalities
in temporal lobe epilepsy
As already indicated, MRS may frequently show
bilateral hippocampal abnormalities (i.e. NAA is
lower than normal control values in both left and
right hippocampi in patients with seizures).
This
could reflect bilateral sclerosis, or it could
represent
the effect of seizure propagation from the
epileptogenic
hippocampus to the other side, causing metabolic
impairment. In this regard, it is interesting to
note
that the metabolism of the contralateral hippocampus
typically “improves” after ipsilateral surgery and
seizure
control, suggesting neuronal dysfunction rather
than irreversible neuronal loss. This effect
seems to occur over a time period of months
following
surgery. Conversely, untreated patients may
show progressive worsening of NAA/Cr ratios overtime.
More recent studies using MRSI with 1 ml nominal
spatial resolution have suggested that the network
of brain regions affected by seizures originating in
the mesial temporal lobe can be mapped by looking
for correlations between metabolite levels in
different regions of the brain. Figure 2 shows an
example
of network connection derived from MRSI; in the TLE
patients in this study, in addition to low NAA/Cr
in ipsi- and contra-lateral hippocampi, NAA/Cr was
also lower than controls in both ipsi- and
contralateral
thalami. Furthermore, ipsi-lateral hippocampal
NAA/Cr values were correlated with contralateral
hippocampi, and ipsi- and contra-lateral thalami and
putamina, suggesting these structures are all
functionally
linked and metabolically affected by seizure
activity.
Seizure activity may also result in more widespread
metabolic abnormalities; for instance, it has been
found that frontal lobe NAA levels are lower in TLE
patients than in controls in both gray and white
matter
regions, as well as in other lobes.
Conversely,
in patients with epilepsy in the neocortex,
hippocampal NAA reductions have also been reported.
These factors should be kept in mind when using
MRSI to evaluate whether seizures are of temporal,
neocortical,
or extratemporal origin; however, in making
this distinction, the largest metabolic abnormality
is generally reported to be in the site of seizure
onset.
Finally, it is interesting to note that many
published
MRS studies are apparently successful in identifying
the epileptogenic temporal lobe, despite the fact
that large regions-of-interest are often used for
spectroscopic
analysis (e.g. 8 cm3 for single-voxel studies).
Voxels are even larger for 31P studies (e.g. ≈ 30
cm3 or
larger). Since the hippocampus occupied only a small
fraction of these localized volumes, these results
might
indicate that there are diffuse spectroscopic
abnormalities
in the temporal lobe, even when the only MRI
finding is that of hippocampal atrophy. These
results
are therefore consistent with the common observation
by PET of extensive hypometabolism throughout the
temporal lobe. Since seizure control is often
obtained
by selective amygdalohippocampectomy, clearly not
all of the metabolically abnormal tissue is
epileptogenic.
 MRS in TLE cases where MRI is normal,
or symmetrically abnormal
MRS in TLE cases where MRI is normal,
or symmetrically abnormal
Arguably, the most useful scenario for MRS is when
results from other modalities (particularly MRI) are
either normal, ambiguous, or bilaterally abnormal.
In these TLE patients, MRS has the potential of
lateralizing
the epileptic focus. Presence of bilateral MRI
abnormalities does not necessarily indicate a poor
surgical outcome, but does increase the
difficulty
in correctly lateralizing the epileptogenic source.
In an
MRSI study of 21 patients with bilateral hippocampal
atrophy who were operated on the side of greatest EEG
abnormality, it was found that factors in favor
of good surgical outcome were: (1) concordant
MRSI EEG
localization; (2) greater asymmetry of NAA/Cr
between hippocampi; and (3) an absence of
contralateral posterior NAA/Cr abnormalities.
MRS may also play a role when MRI is normal – in
a study of 7 patients with intractable epilepsy but
completely normal MRI findings, it was found that
5 of 7 cases had abnormal NAA/(Cr+Cho) ratios, 2
of which were bilateral. Although this study did
not report detailed EEG correlation or surgical
outcome,
it did suggest that MRS may provide additional
information when MRI is normal. NAA has also been
found to be lower than normal control values in the
ipsilateral hippocampus (to EEG) in another study of
MRI negative patients. However, another study
found that metabolic abnormalities (in particular,
well localized
NAA asymmetry) surprisingly did not predict
seizure-free outcome after surgery, although the
presence of contralateral abnormality did predict
poor outcome in this group.
 Cortical malformations
Cortical malformations
There have been a number of reports of MRS in
patients with malformations of cortical development
(MCD). Despite their frequent
epileptogenic
nature, MCDs typically show only subtle (or
sometimes no) metabolic abnormalities;
when metabolic abnormalities are observed, most
commonly
NAA is reduced and Cho increased, particularly
for focal cortical dysplasias. As with other types
of
epilepsy, metabolic changes remote from the presumed
seizure focus (e.g. in the contralateral hemisphere)
may
be different (typically lower NAA/Cr) from healthy
controls, and in fact were not significantly
different
from the ipsilateral side.
 Frontal lobe epilepsy
Frontal lobe epilepsy
There have been fewer reports of MR spectroscopy
in extratemporal epilepsy than in TLE. Garcia et al.
have studied frontal lobe epilepsy using both 31P
and 1H MR spectroscopy. In the proton study, all
eight cases exhibited a reduced NAA/Cr ratio in the
epileptogenic tissue compared to an anatomical
similar
contralateral location. Stanley et al. also reported
the results of proton spectroscopy imaging in 20
cases
with frontal lobe epilepsy. As in TLE, it was
found
that the ratio of NAA/(Cho+Cr) successfully
lateralized
the epileptogenic tissue as defined by EEG.
Widespread NAA reductions were also noted (i.e. the
contralateral NAA was also lower than control
values),
indicating extensive neuronal loss not confined to
just the side of seizure onset, as commonly observed
in TLE.
 Childhood epilepsies, Rasmussen’s
encephalitis
Childhood epilepsies, Rasmussen’s
encephalitis
A number of epilepsies of childhood have been
studied
by MRS. MRS has also been used to
investigate
cerebral metabolism in the “ketogenic diet”,
which is becoming increasingly popular as an
alternative
to pharmacological means of seizure control –
using 31P MRS, improvements in bioenergetic status
have been reported while proton
MRS has shown that the ketone bodies such as β-hydroxy-butyrate
and acetone may be detected.
Rasmussen’s encephalitis (RE) is a rare, chronic,
and progressive epilepsy of childhood involving one
hemisphere of the brain. While the cause is largely
unknown, currently the only treatment that can
reliably
provide effective seizure relief is hemispherectomy.
Definitive diagnosis is usually made on the basis of
clinical, electroencephalographic, and neuroimaging
findings; however, in the early stages diagnosis may
not be straightforward. Breiter et al. found
hemispheric
NAA reductions in 5 cases of Rasmussen’s syndrome, MRSI the whole hemisphere
shows low NAA, consistent with neuronal loss. It can
also be seen that choline is elevated in the
affected
hemisphere, particularly the white matter,
consistent
with the microglial proliferation typically seen on
pathology. The hemispheric nature of the metabolic
involvement confirms the clinical observation that
complete hemispherectomy is necessary for effective
seizure control in most cases.
An alternative pattern of involvement shows a more regional
distribution,
with high Cho and low NAA primarily occurring
in the insular cortex, putamen, and frontal lobe.
 Neurotransmitters: brain GABA levels
Neurotransmitters: brain GABA levels
While the majority of MRS studies of epilepsy
have studied the most readily observed metabolites
Cho, Cr, and NAA, by the use of spectral editing
methods it is also possible to measure the
inhibitory
neurotransmitter, γ-aminobutyric acid (GABA). Typically, macromolecules and the
dipeptide homocarnosine also co-edit with GABA, so
that the peak observed by MRS is sometimes labeled
GABA+ to distinguish it from “pure” GABA. It has
been reported that GABA may be (globally) decreased
in the brains of epilepsy patients with poor seizure
control, and that GABA levels can be increased
(and seizure control obtained) using the
antiepileptic
drugs vigabatrin, topiramate,] and gabapentin. Techniques of this type are promising for
monitoring
the effects of therapy and establishing optimal
drug dosages, and will be more widely used as
the
MEGA-PRESS editing technique becomes commercially
available. It should be noted that other compounds
of potential clinical significance, such as
glutamate and N-acetyl-aspartyl-glutamate (NAAG) can
also be measured using MEGA-PRESS.
 Some recommendations for MRS
protocols for patients with epilepsy
Some recommendations for MRS
protocols for patients with epilepsy
Since metabolic changes in interictal epilepsy
patients
are often subtle, high quality MRS with good SNR is
essential. Ideally, high-field field scanners (e.g.
3 T) with
multiple phased-array receiver coils should be used.
For temporal lobe epilepsy, the simplest protocol
is to compare the body of the left and right
hippocampi
at intermediate TE (typically 140 msec) using
single-voxel PRESS, or PRESS-MRSI, angulated along
the long axis of the hippocampus. While this may seem
relatively simple, in fact considerable care has to
be
taken with these protocols. Voxels should be
positioned
carefully (both because of the small structures
to be observed, and also because of metabolic and
field
homogeneity changes along the hippocampus),
preferably using full 3-view localizers (axial
T1-weighted
3D “MP-RAGE” scan reconstructed in both sagittal
and coronal views works well. High bandwidth
slice-selective RF pulses are preferable to
minimize left–right asymmetries due to chemical
shift displacement effects. Second-order shimming
is also important (especially at higher fields such
as 3 T), particularly for MRSI protocols, since
significant
non-linear field inhomogeneities occur in the
temporal lobes which cannot be corrected using
linear
shims alone. At the anterior tip of the temporal
lobe (pes or head of the hippocampus), field
homogeneity
is usually particularly poor because of
susceptibility
effects from the nearby paranasal sinuses.
Poor field homogeneity results in insufficient
quality
spectra for analysis in most adult subjects, even
with
high order shimming corrections. This is
unfortunate,
since this is often the target of surgical resection
when anterior mesial temporal lobectomies are
performed, and is presumably the primary site of
pathology. Because of the poor field homogeneity in
anterior regions, as well as potential lipid
contamination
from retro-orbital fat and other skull base
structures, it is helpful to apply saturation bands
in
these anterior regions.
For extratemporal lobe epilepsy, the site of the
epileptogenic focus may be unknown, or even when
known (e.g. MCDs) the spectroscopic findings may
be heterogeneous, therefore in these cases the best
approach is to use MRSI with high spatial coverage
(e.g. multi-slice or 3D). MRSI is also important
since
the observation of abnormal metabolism remote from
(e.g. surrounding, connected via fiber pathways, or
contralateral to) the primary focus is also commonly
reported, and may be of clinical significance.
Since MRS changes in epilepsy are generally
subtle, demands are placed on the accuracy of both
the acquisition technique and spectral analysis
software methods. For single-voxel epilepsy MRS,
the LC model software is particularly recommended,
since it can provide metabolite concentrations and
yield uncertainty estimates. In addition, it is
advisable to have matched control data using the
same scanner, brain region, and MRS technique for
comparison; more advanced studies use statistical
tests (such as a z-score) to estimate how abnormal
any particular spectrum may be. Precision will
also be improved by applying corrections to
metabolite
concentrations according to the gray matter,
white matter, and CSF composition within the
localized voxel.
 Conclusions
Conclusions
In summary, MRS of epilepsy is now a relatively
mature field, with the reduction of NAA in abnormal
tissue the most common finding. Despite this
observation,
and the relatively high reported sensitivity and
specificity of MRS for seizure focus lateralization
in
TLE in most research studies, the technique has not
found widespread application in clinical practice.
This
is due to the reasons that are listed in the
introduction,
of which the most likely is the relatively subtle
metabolic
changes that are found in most patients. Such
subtle changes, along with data that are susceptible
to
minor instrumental imperfections, make
interpretation
of individual studies challenging. The “added-value”
of MRS compared to other diagnostic techniques
remains questionable, although it is apparent that
it may be helpful in at least some of the cases that
are
MRI negative or symmetrically abnormal. As high field
scanners and proton MRS techniques improve
(i.e. with improved SNR and accuracy of quantitation),
and as the use of editing techniques for
neurotransmitters
such as GABA or glutamate increases, it
is hoped that the clinical utilization of MRS in the
evaluation of patients with epilepsy will increase.
In
the long run, MRS may help obviate the need for
invasive EEG procedures and expensive alternative
imaging procedures such as PET.
|
Several cases of epilepsy patients |
 Case of PGE with bitemporal affection with no
morphological data supporting mesial sclerosis, but
the NAA is decreased in both mediobasal temporal
lobes with no noticeable asymmetry.( Fig.1,2,3,4,5)
Case of PGE with bitemporal affection with no
morphological data supporting mesial sclerosis, but
the NAA is decreased in both mediobasal temporal
lobes with no noticeable asymmetry.( Fig.1,2,3,4,5) |